Meaningful Progress in the Treatment of Breast Cancer
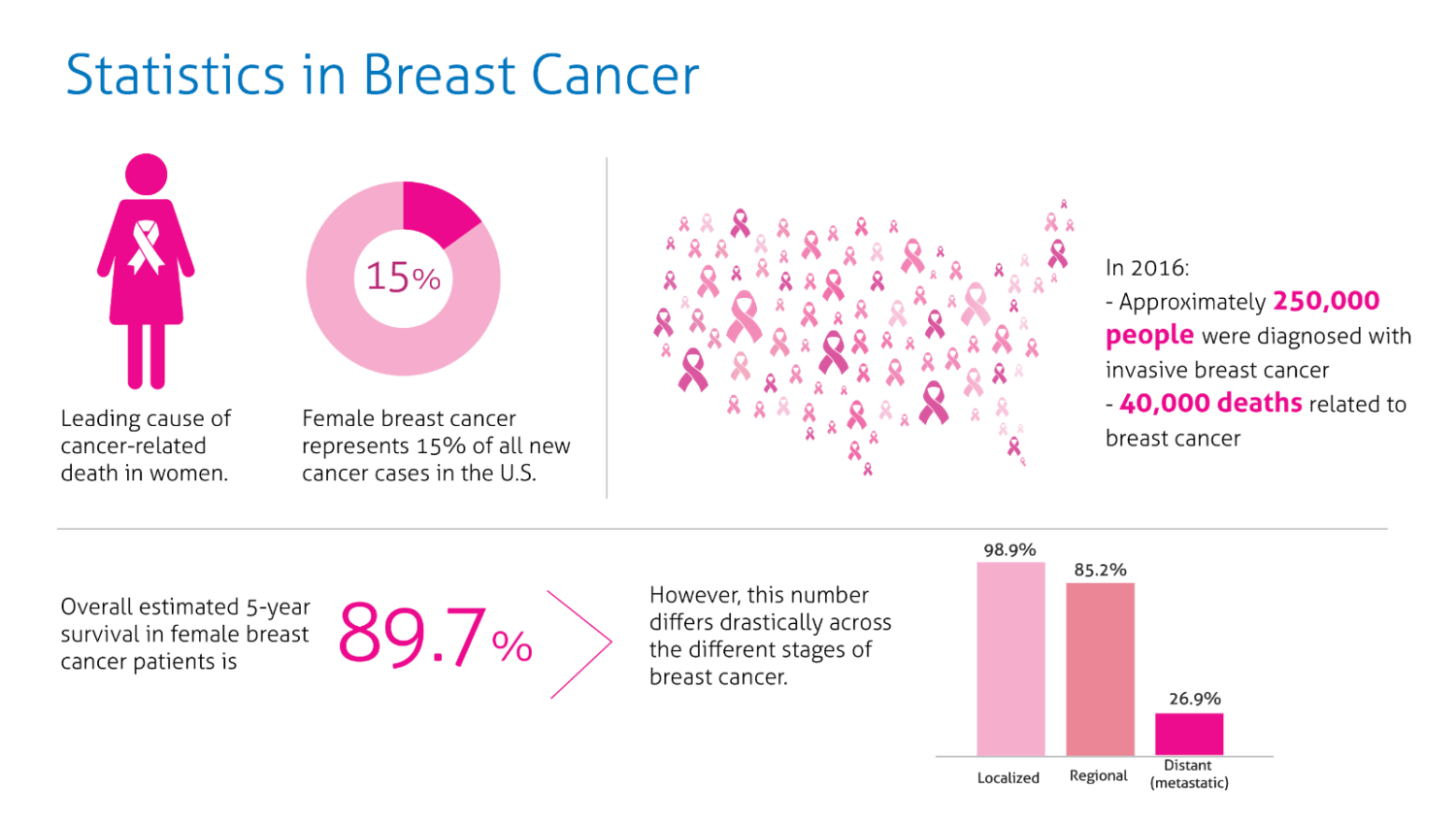
Breast Cancer Awareness Month is a chance to raise awareness and remind each other about the importance of early detection. When breast cancer is detected early, and is in the localized stage, the 5-year relative survival rate is 100%.
For those in need of treatment, several new therapies have been approved by FDA for breast cancer just this year, including a new wave of immunotherapies and combination therapies, as well as promising new treatments for triple-negative breast cancer.
- Trastuzumab and hyaluronidase-oysk injection (HERCEPTIN HYLECTA™, Genentech), a monoclonal antibody combined with an enzyme to enhance uptake, was approved for the treatment of HER2 overexpressing breast cancer in February 2019. This novel treatment is administered subcutaneously.
- Atezolizumab (TECENTRIQ®, Genentech), a monoclonal antibody, in combination with nab-paclitaxel (ABRAXANE®) chemotherapy, was approved for PD-L1 positive or metastatic triple-negative breast cancer in March 2019. This was the first FDA-approved regimen for breast cancer to include immunotherapy.
- Ado-trastuzumab emtansine (KADCYLA®, Genentech), an antibody-drug conjugate that combines Herceptin and the chemotherapy medicine emtansine, was approved for the adjuvant treatment of patients with HER2-positive early breast cancer in May 2019.
- Alpelisib (PIQRAY®, Novartis), a PI3K inhibitor in combination with fulvestrant for postmenopausal women, and men, with HR-positive, HER2-negative, PIK3CA-mutated, advanced or metastatic breast cancer, was approved in May 2019.
Just last weekend, on the doorstep of Breast Cancer Awareness Month, Merck presented interim data from the Phase III KEYNOTE-522 trial in patients with early-stage triple-negative breast cancer at the ESMO 2019 Congress in Barcelona.
The trial investigated neoadjuvant Keytruda plus chemotherapy, followed by adjuvant Keytruda for triple-negative breast cancer. The combination of Keytruda and chemotherapy resulted in a statistically significant increase in pathological complete response (i.e. no invasive residual cancer in breast and lymph nodes) versus chemotherapy alone – from 51.2% to 64.8% with the neoadjuvant combination.
On the alternative medicine front, recent research published in the International Journal of Molecular Sciences has hypothesized the ability of cannabinoids to reduce angiogenesis and tumor metastasis in breast cancer models. Stay tuned!
With continued early detection practices and a broader range of treatment options, we look forward to a future in which we can prevent, detect, treat or manage this disease.
As your biopharma customers develop these ever-improving cancer medicines, you want to be there to support them with your products and services. If you have questions on how to adapt your business to engage them, raise your brand awareness, or increase your lead generation, fill out the form below and we’ll be in touch very soon.
Sign Up
Stay connected with Hapatune and receive bioprocess market trends, marketing insights, industry tidbits and Hapatune happenings.